Ever wondered what makes a balloon float, or why your soda fizzes? The secret lies in the fascinating world of gas properties! And guess what? You can actually play around with these concepts in a super fun and interactive way through a Gas Properties Simulation Activity. Think of it as a virtual playground for understanding how gases behave, and the best part is, we've got the answer key to help you conquer it!
Why is This So Cool?
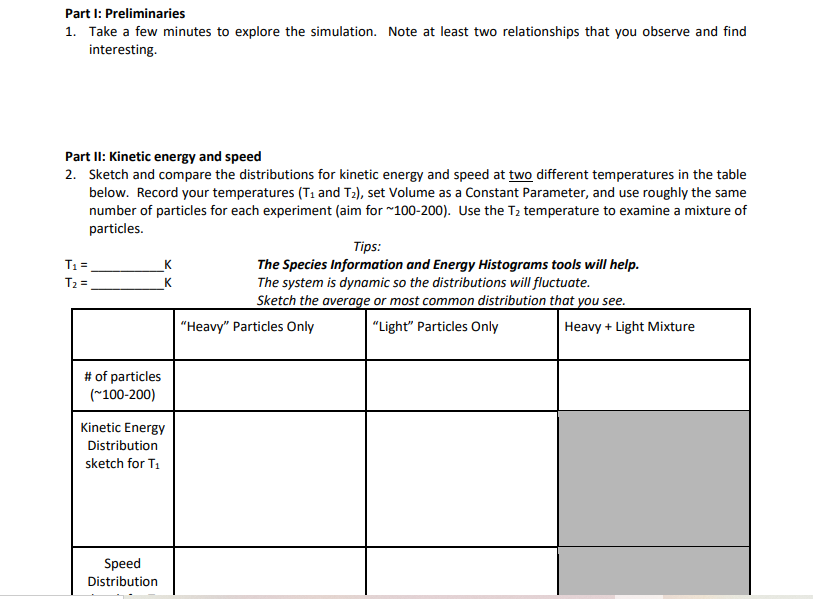
Science doesn't have to be confined to dusty textbooks. The beauty of a simulation activity is that it brings abstract ideas to life. You get to manipulate variables like temperature, pressure, and volume, and immediately see the results. It’s like being a mad scientist in your own virtual lab, but without any of the mess! Understanding gas properties is fundamental to so many things around us, from the weather patterns we experience daily to the advanced technologies that power our world. This activity makes learning about these essential concepts engaging and, dare we say, exciting!
What's the Goal Here?
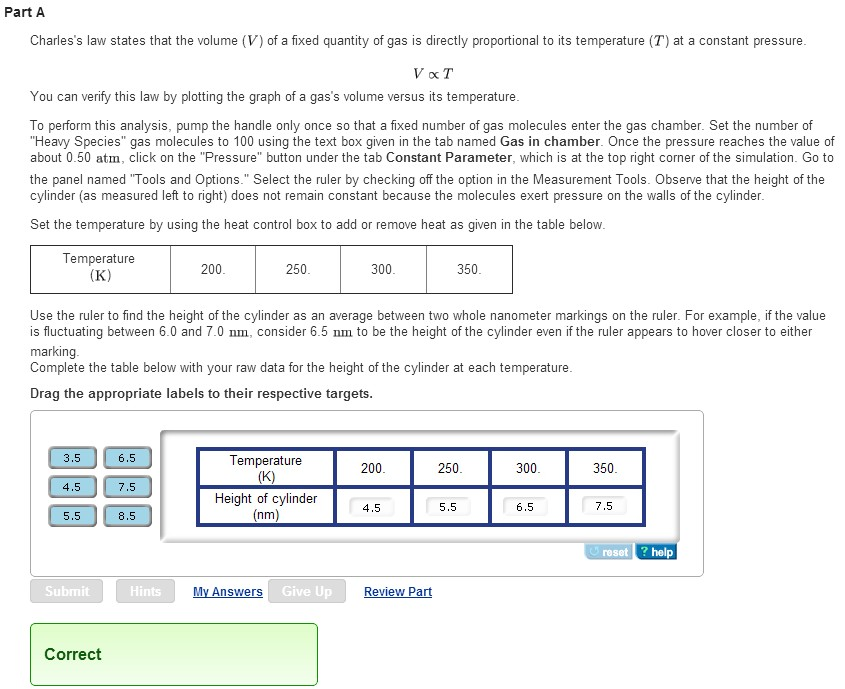
The primary purpose of a Gas Properties Simulation Activity is to provide a hands-on, visual experience that solidifies your understanding of the fundamental laws governing gases. You'll get to explore concepts like Boyle's Law, Charles's Law, and the Ideal Gas Law. These laws describe the relationships between pressure, volume, temperature, and the amount of gas. By playing with the simulation, you'll discover how changing one of these factors affects the others.
The benefits are immense. You'll develop a deeper intuition for how gases work, making it easier to grasp more complex scientific principles later on. Plus, it’s a fantastic way to develop critical thinking skills. You’ll be observing, hypothesizing, and analyzing data, just like a real scientist. The simulation allows you to conduct experiments that might be difficult or impossible in a traditional classroom setting, offering a safe and repeatable environment for learning.
Unlocking the Secrets with the Answer Key
Now, about that answer key! Think of it as your trusty guide through the simulation's challenges. It’s not about cheating; it’s about understanding. The answer key provides the correct responses to specific questions or tasks within the activity. This is incredibly useful for:

- Checking your work: Did you correctly predict how increasing the temperature would affect the volume? The answer key confirms it.
- Understanding misconceptions: If you get something wrong, the answer key can highlight where your understanding might need a little tweaking. It helps you identify what you thought happened versus what actually happens according to scientific principles.
- Reinforcing learning: Seeing the correct answers and connecting them back to the simulation’s outcomes reinforces the concepts you've learned. It’s like a quick win that boosts your confidence.
- Guiding your exploration: Sometimes, the answer key can hint at the underlying principles you should be focusing on, encouraging you to look for specific relationships within the simulation.
The beauty of using an answer key for a simulation is that it complements the active learning process. You engage with the simulation first, making your own observations and predictions. Then, you consult the answer key to verify your findings and deepen your comprehension. It’s a dynamic way to learn, where you’re actively involved in the discovery process.
Let's Dive In!
Imagine you’re in the simulation, and you decide to increase the temperature of a gas in a container. What do you think will happen to the pressure if the volume stays the same? According to Gaye-Lussac's Law (a close cousin to Charles's Law!), the pressure will increase because the gas particles are moving faster and hitting the walls of the container more frequently and with more force. The answer key would confirm this, and perhaps even offer a brief explanation to solidify your understanding.
Or, consider Boyle's Law. If you decrease the volume of a container holding a gas at a constant temperature, what happens to the pressure? The answer key would tell you that the pressure increases. Why? Because the gas particles have less space to move around, so they collide with the container walls more often.
The Ideal Gas Law, which combines these relationships, is also a star player in these simulations. It’s expressed as PV = nRT, where P is pressure, V is volume, n is the amount of gas (in moles), R is the ideal gas constant, and T is temperature. The simulation allows you to see how these variables are interconnected. If you increase the temperature (T) while keeping the amount of gas (n) constant, and you want to keep the pressure (P) the same, you'll have to increase the volume (V).
Using the Gas Properties Simulation Activity Answer Key transforms a potentially confusing topic into an accessible and enjoyable learning experience. It's your passport to understanding the invisible forces that shape our world, all through the magic of interactive science!