Hey there, fellow curious minds! Ever looked at something and thought, "Hmm, it's not quite what it seems?" Like when you're trying to describe a friend who’s a little bit of everything – sometimes super outgoing, other times perfectly content with a quiet night in? Well, buckle up, because we’re about to dive into something similar, but with molecules! Today, we’re chatting about formal charges and how they help us understand those quirky things called resonance structures in a molecule called HCNO. Sounds fancy, right? But trust me, it’s more like a fun puzzle than a scary chemistry exam.
So, what’s this HCNO all about? Think of it as a tiny molecule, a building block of sorts. It’s got a hydrogen (H), a carbon (C), a nitrogen (N), and an oxygen (O) all holding hands (or rather, sharing electrons). Now, these atoms are a bit like artists with different palettes. They can arrange themselves and share their electron paint in a few slightly different ways, creating what we call resonance structures. Imagine a really good baker who can make the same cookie dough into several different shapes – a star, a circle, maybe even a little pretzel. They’re all fundamentally the same dough, but they look a bit different.
Now, the big question is: which of these "shapes" is the real deal? Or are they all kind of real? This is where our superhero, formal charge, swoops in to save the day. Think of formal charge as a way to assign a "score" to each atom in a Lewis structure (that's just a fancy drawing of how the atoms are connected and share electrons). It helps us figure out where the electrons prefer to hang out, or rather, which arrangement is the most stable and likely.
Why should you even care about this molecular guesswork? Well, understanding these resonance structures and their formal charges is like knowing the secret ingredient that makes a dish taste amazing, or figuring out why your favorite song just feels right. It helps us predict how molecules will behave, how they’ll react with other molecules, and why certain materials have the properties they do. It’s the nitty-gritty that explains the big picture!
The Many Faces of HCNO: A Closer Look
Let’s get a bit more specific with our little HCNO. This molecule can exist in a few different ways, or resonance forms. Imagine you have a group of friends deciding where to go for dinner. One friend might suggest a fancy Italian place, another a cozy Thai restaurant, and a third a casual burger joint. They’re all possibilities, and depending on what everyone’s in the mood for, one might be chosen. Similarly, HCNO can be drawn with its atoms connected in different ways, leading to different distributions of electrons. We’ll focus on the most common and interesting ones.
Resonance Form 1: The "Balanced Budget" Approach
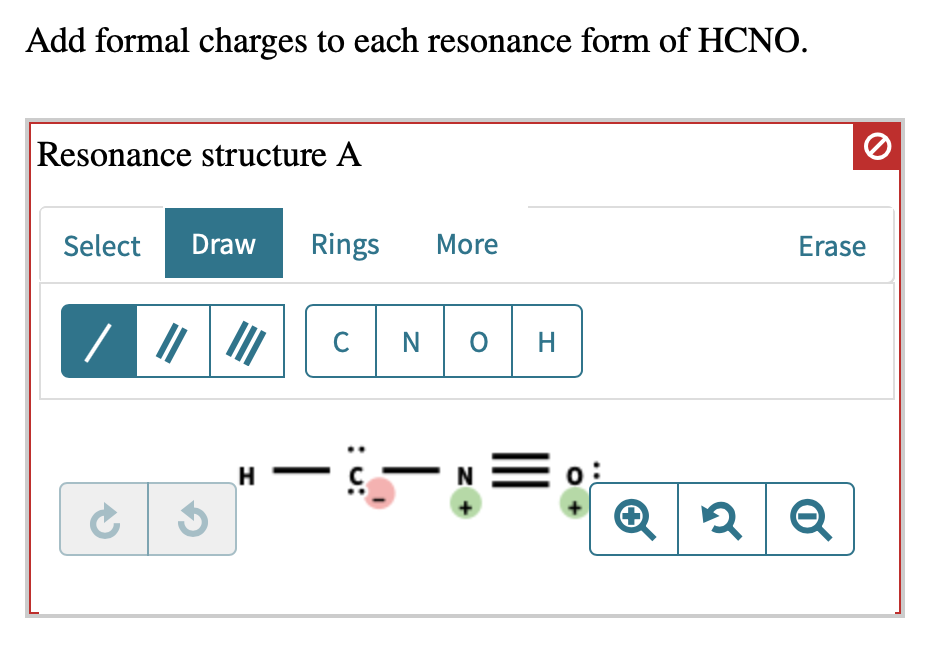
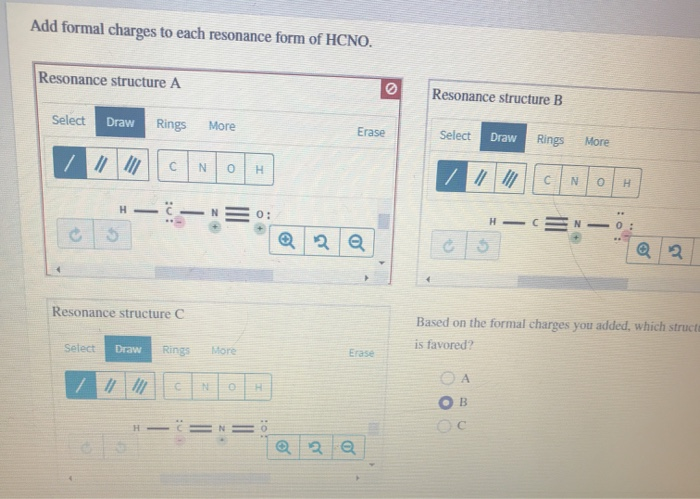
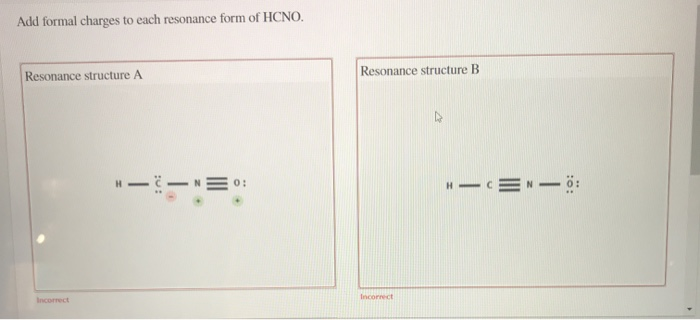
In one common way HCNO can arrange itself, we have a structure that looks something like this: H-C≡N-O. (Don't worry about memorizing this; just visualize the connections!). In this setup, let’s play the formal charge game. Remember how we assign scores? We count the electrons around each atom in our drawing and compare it to how many electrons that atom should have when it's all by its lonesome. It’s like checking your bank account: are you spending more than you have, or are you living within your means?

For this specific arrangement (H-C≡N-O), we’d find that the hydrogen atom has a formal charge of 0. The carbon atom also has a formal charge of 0. The nitrogen atom, however, might end up with a formal charge of 0 as well. And the oxygen? You guessed it, often a formal charge of 0. This is like a perfectly balanced budget – everyone’s happy, no one's overspending or underspending on electrons. This is generally a very good thing in the molecular world, because atoms that have a formal charge of zero are usually more stable.
Think about it like this: if you have a perfectly organized desk, where everything is in its right place, you’re probably going to be more productive and less stressed. Molecules with formal charges of zero are like that tidy desk – they’re more content and less likely to go looking for trouble (or, in chemistry terms, react wildly).
Resonance Form 2: The "Slightly Overdrawn" Scenario
But wait, there’s another way HCNO can be drawn! Let’s try H-C=N=O. Again, visualizing the connections is key. Now, let’s do our formal charge math again. This time, things might get a little more interesting. The hydrogen is still happily at 0. The carbon might be at 0. But the nitrogen? It could end up with a formal charge of +1! And the oxygen? It might have a formal charge of -1.

This is like having one friend who’s always willing to pick up the tab (+1, the giver) and another who’s always happy to let them (+1, the receiver, but this is a bit more complex). In this structure, we have a positive charge on the nitrogen and a negative charge on the oxygen. This might sound a bit chaotic, like someone who’s a little overdrawn at the bank. But here’s the cool part: even with these charges, this resonance form contributes to the overall picture.
Why? Because the charges are on atoms that can handle them. Nitrogen, being a bit electronegative (meaning it likes to attract electrons), can handle a positive charge better than, say, a hydrogen. And oxygen, being even more electronegative, is perfectly happy to carry a negative charge. It’s like putting a heavy box on a strong person’s shoulders – they can manage it. This arrangement, despite having formal charges, can still be a significant contributor to the molecule’s true identity.
Resonance Form 3: The "Borrowing from Peter to Pay Paul" Situation
Let's consider one more possibility: H-O-C≡N. This is another way to connect these atoms. Time for our formal charge detective work! Hydrogen: 0. Oxygen: -1. Carbon: +1. Nitrogen: 0.

Here, the oxygen is carrying a negative charge, and the carbon is carrying a positive charge. This is like one friend lending money to another, who then lends it to a third friend. It’s a bit of a juggling act. Again, the charges are on atoms that can somewhat tolerate them. Oxygen can handle a negative charge, and carbon can handle a positive charge, especially when it's adjacent to a very electronegative atom like nitrogen.
However, in this specific case, the formal charges are on atoms that are generally less favorable for them. Oxygen generally prefers to have a negative charge than a positive one, and carbon is usually happiest neutral. This makes this resonance form a bit like a hesitant dance – it’s possible, but maybe not the smoothest performance.
Putting it All Together: The Real HCNO
So, we’ve got these different "drawings" or resonance forms. The key takeaway is that the actual HCNO molecule isn't just one of these drawings; it's a blend of all of them. Imagine a really vibrant painting. It’s made up of countless tiny brushstrokes of different colors. The overall impression isn't just one color; it's the harmonious combination of all of them.

When we calculate formal charges, we’re essentially trying to find the most "comfortable" or "stable" arrangement. Molecules, like us, tend to gravitate towards the easiest, most energy-efficient way of being. The resonance form with the fewest formal charges, and where any charges are on the most appropriate atoms, is usually the most important contributor to the molecule's true structure. In the case of HCNO, the structure with all zero formal charges is often considered the most significant.
This is why understanding formal charges is so handy. It's our guide to figuring out which resonance structure is the "main character" and which are the "supporting cast." It helps us understand the electron density – where the electrons are most likely to be found – which, in turn, tells us how the molecule will interact with its environment. It’s like knowing your friend’s moods: sometimes they’re bubbly, sometimes they’re thoughtful, and understanding these different states helps you know how to interact with them best.
So, the next time you hear about formal charges and resonance structures, don't let it intimidate you. Think of it as a fun way to peek into the secret lives of molecules, a puzzle that helps us appreciate the incredible complexity and beauty of the chemical world. And who knows, it might even make you appreciate the little quirks of your own social circle a little more!