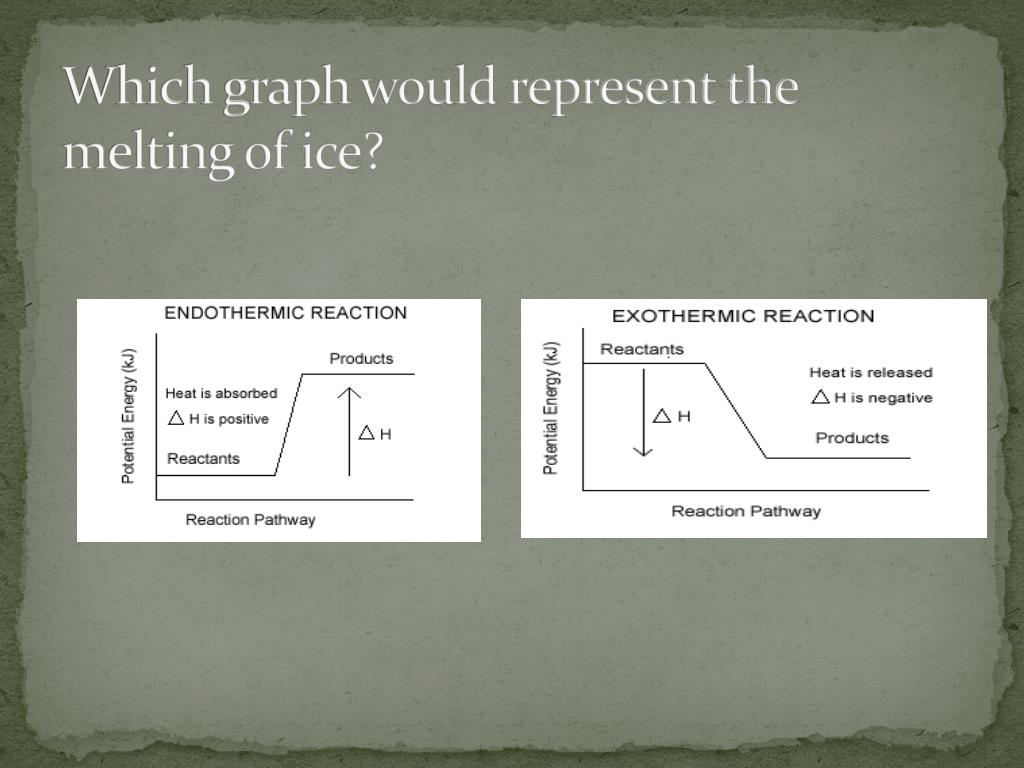
Hey there, science adventurers! Ever seen that spooky, foggy stuff at Halloween parties or in science class? You know, the stuff that makes your drinks look like they’re brewing magic potions? That’s dry ice, and it’s way cooler than it looks (literally!). Today, we're diving into a little secret about this frosty wonder: does it get colder when it disappears, or does it warm things up? Get ready for some seriously fun science!
Imagine you’ve got a big, frosty block of dry ice. It’s sitting there, looking all mysterious and solid. But here’s the kicker: it’s not actually melting into a puddle like your ice cream on a hot day. Nope! Dry ice does something far more dramatic. It goes straight from being a solid, frozen chunk to a gas, skipping the whole liquid phase. This is called sublimation, and it’s like a magician pulling a disappearing act right before your eyes!
Now, the big question: when this magical dry ice vanishes into thin air, what’s happening to the temperature around it? Is it like a mini-refrigerator, making everything even colder as it goes? Or is it more like a tiny, invisible fireplace, spreading warmth as it fades away? Prepare yourselves, because the answer is going to blow your cool, frosty minds!
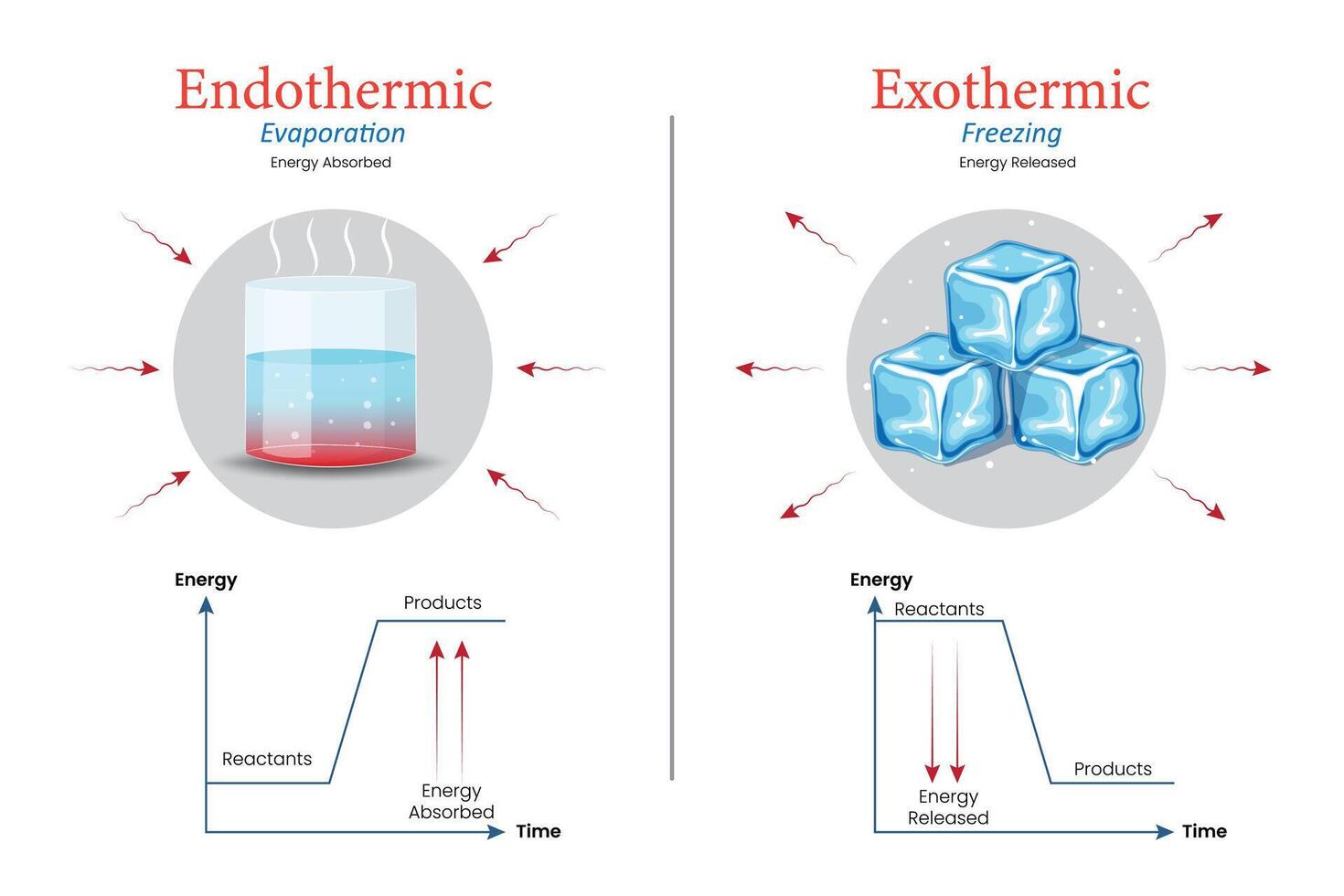
When dry ice goes through its disappearing act, it’s actually gobbling up heat. Yes, you heard that right! It’s a heat-hoover, a temperature-thief, a cold-capturing champion! It needs energy to perform its amazing transformation from solid to gas, and where does it get that energy? From its surroundings! Think of it like this: you’re feeling a bit sleepy and chilly, so you snuggle up under a blanket. The blanket is like the dry ice, and your body heat is the energy it’s absorbing to keep you warm. Except, in the case of dry ice, it’s absorbing the heat from the air, the table, and anything else nearby. This process of absorbing heat from its environment is called endothermic. So, while the dry ice itself is incredibly cold (we’re talking a chilling -78.5 degrees Celsius, or -109.3 degrees Fahrenheit!), the act of it disappearing makes its immediate surroundings feel a bit… well, less frigid than they would be without it. It’s like it’s stealing the warmth to fuel its grand escape!
Let’s picture this with something we all understand: a cold drink on a hot day. You put a cube of regular ice in your lemonade, and it melts, making your lemonade colder. Pretty straightforward, right? But now, imagine you could make your lemonade even more dramatically chilled, and it would vanish without leaving a watery mess! That’s the power of dry ice. If you were to toss a little bit of dry ice into a bowl of lukewarm water, you’d see those amazing fog effects. That fog is actually water vapor condensing because the air around the disappearing dry ice is getting really cold. The dry ice is busy snatching up all the heat it can find, creating that super-chilly environment. It’s like it’s saying, "Out of my way, heat! I’ve got a transformation to complete!"
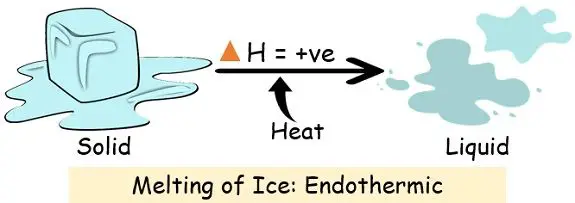
So, the vanishing act of dry ice is an endothermic process. It needs to absorb heat to turn into that ghostly gas we love to watch.
Now, let’s have a little fun with the opposite idea. What if dry ice released heat when it disappeared? Imagine tossing a block of this stuff into a room, and instead of getting colder, the room suddenly felt like a cozy fireplace. That would be a completely different kind of magic, wouldn’t it? That would be an exothermic reaction, where heat is given off. But thankfully (or maybe slightly disappointingly for those who wanted a built-in heater!), dry ice is not playing that game. It’s all about the heat absorption. It’s the ultimate cool customer, and it proves it by drawing warmth towards itself.
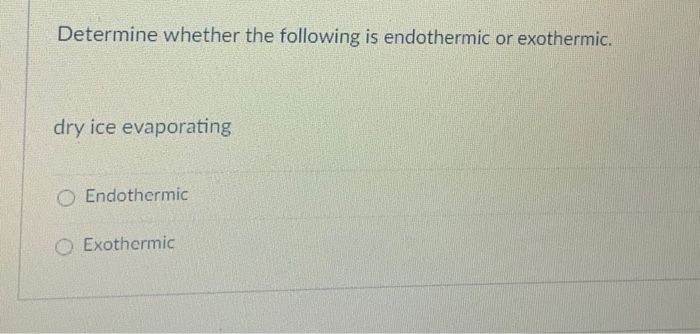
Think about it like a superhero. Dry ice is our superhero of chilling! Its superpower is sublimation, and its secret move is being endothermic. It swoops in, snatches up all the surrounding heat, and BAM! It transforms into a cloud of mystical fog, leaving behind a slightly cooler environment than if it hadn't been there to begin with. It’s not making things extra cold by releasing something; it’s creating the effect of extreme cold by taking away the warmth.
So, next time you see that spectacular fog swirling from a drink or a stage effect, you can impress your friends with your newfound knowledge. You can confidently declare, with a knowing wink, "Ah yes, that’s dry ice! And its vanishing act? It's endothermic – a true heat-seeker!" It’s a simple concept, but it’s at the heart of some of the most visually stunning and scientifically fascinating phenomena. Keep exploring, keep questioning, and keep enjoying the amazing world of science – it’s full of surprises, and a whole lot of fun!