Ever wondered what happens in the microscopic world? It's a place buzzing with activity, even when things seem to be still on the surface. One of the most fascinating concepts in this tiny realm is diffusion, and a question that often pops up is: do molecules stop moving when diffusion stops? It sounds like a trick question, right? If diffusion is about things spreading out, and they've spread out as much as they can, does that mean they just… freeze?
The answer, as it turns out, is a resounding no! This is where things get really fun and a little mind-bending. Diffusion might stop, meaning there's no net movement from one area to another, but the individual molecules themselves are far from taking a nap. Think of it like a crowded room at a party. Initially, people are milling about, moving from one conversation to another, spreading out. Eventually, everyone finds a spot, and the overall movement of the crowd might settle down. But are the people in those spots standing perfectly still? Absolutely not! They're still talking, gesturing, perhaps taking a sip of their drink – they're still moving, just not in a way that changes the overall distribution of people in the room.
The Tiny Dancers of Equilibrium
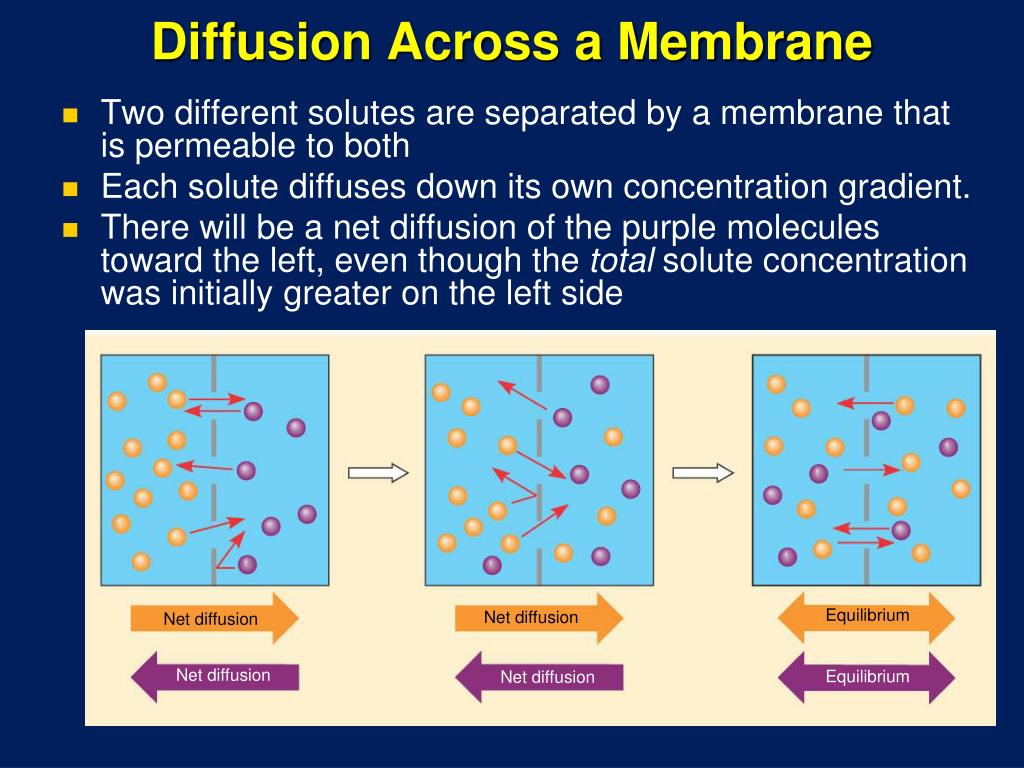
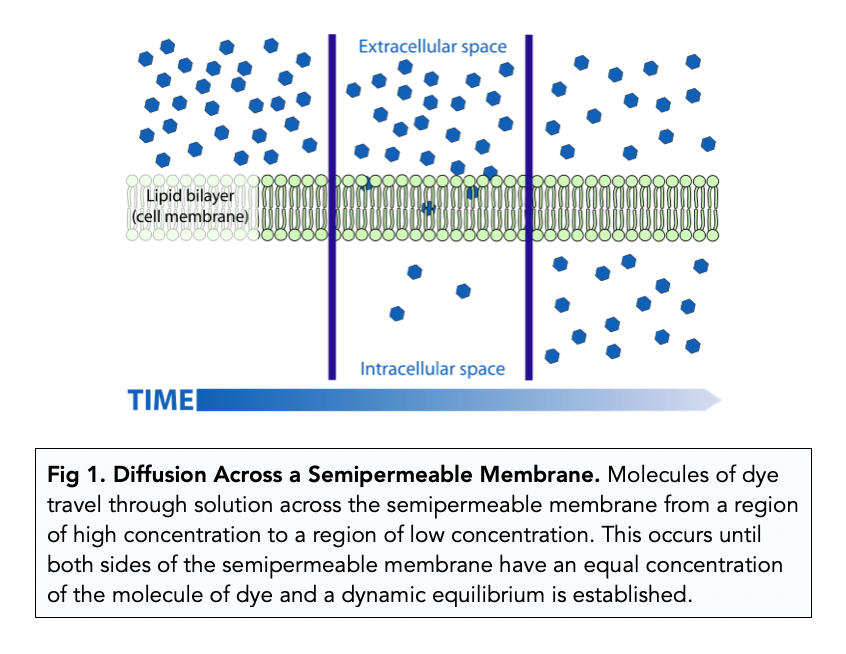
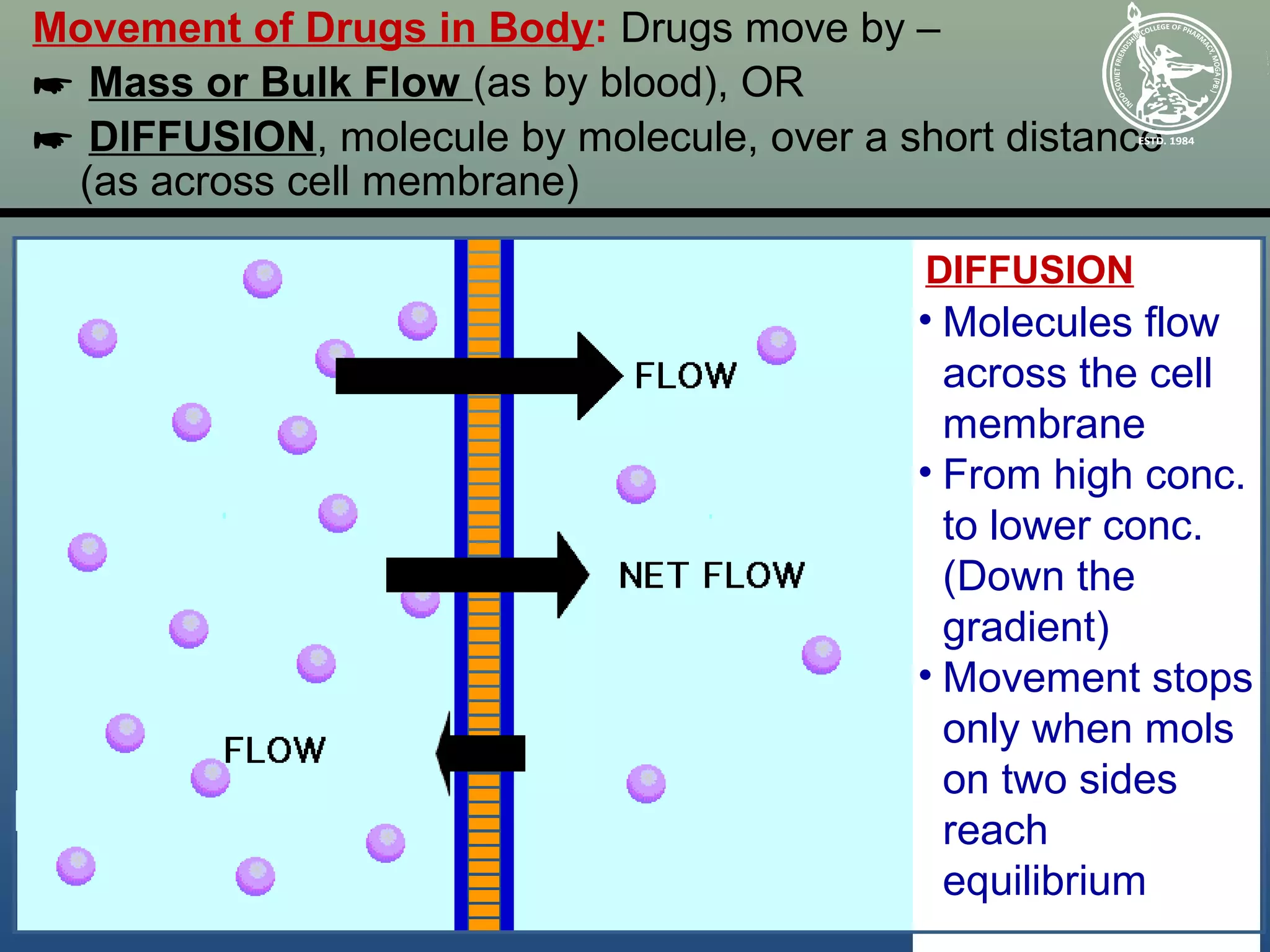
So, what's actually going on when diffusion ceases? We've reached a state of equilibrium. This is a fancy word for balance. In the context of diffusion, equilibrium means that the concentration of molecules is uniform throughout the space. For example, if you put a drop of food coloring in a glass of water, diffusion happens as the coloring spreads out. Eventually, the water will be uniformly colored. At this point, diffusion has stopped, but the color molecules are still zipping around randomly. They bump into water molecules, change direction, and generally have a grand old time.
The key here is that while there's no net movement in any particular direction, the movement of individual molecules is continuous and random. Imagine a single molecule of food coloring. It's not like it decides, "Okay, I've reached my final destination, I shall now remain stationary." Instead, it's constantly being jostled and knocked about by the surrounding water molecules. This random motion, known as Brownian motion, is a fundamental characteristic of matter at the molecular level. Even at equilibrium, these tiny dancers continue their perpetual waltz.

Why This Matters (Besides Being Cool!)
Understanding that molecules don't stop moving at equilibrium isn't just a fun fact for trivia night; it has significant implications in various scientific fields. For instance, in biology, cells rely on diffusion for many essential processes, like transporting nutrients in and waste products out. When these transport processes reach equilibrium, it doesn't mean the cell stops functioning. The molecules involved are still moving, ensuring the dynamic balance necessary for life.
In chemistry, equilibrium is a crucial concept for understanding chemical reactions. Many reactions are reversible, meaning they can proceed in both forward and reverse directions. At equilibrium, the rate of the forward reaction equals the rate of the reverse reaction. This doesn't mean the reaction has stopped; it means that for every molecule that reacts in one direction, another molecule reacts in the opposite direction, resulting in no change in the overall concentrations of reactants and products. This delicate balance is maintained by the constant, ongoing movement and interaction of molecules.

Even in everyday phenomena, this principle is at play. Think about the smell of baking cookies wafting through your house. Initially, the scent molecules are concentrated near the oven, and diffusion spreads them outwards. When the scent seems to have permeated the entire house, diffusion has largely ceased, but the aroma molecules are still actively moving and bumping into air molecules. Your nose detects them because of this persistent, random motion. So, the next time you see something that appears to be perfectly still or uniformly distributed, remember the incredible, ceaseless dance of molecules happening beneath the surface. They are the unsung heroes of our seemingly static world, forever in motion!
The universe is not only stranger than we imagine, it is stranger than we can imagine. - J.B.S. Haldane